Case Study: Dehisced leg trauma wound, skin grafts
The Avance Solo Negative Pressure Wound Therapy System is indicated for patients who would benefit from wound management via the application of negative pressure, particularly as the device may promote wound healing through the removal of exudate, infectious material.
Photographs and case notes kindly supplied by Sanna Kouhia, Vascular Surgeon; Tuula Eskelinen, Wound Care Nurse; Arja Korhonen, Wound Care Nurse, the surgery outpatient clinic / wound outpatient clinic, Kainuu Central Hospital, Kajaani, Finland
Wound history
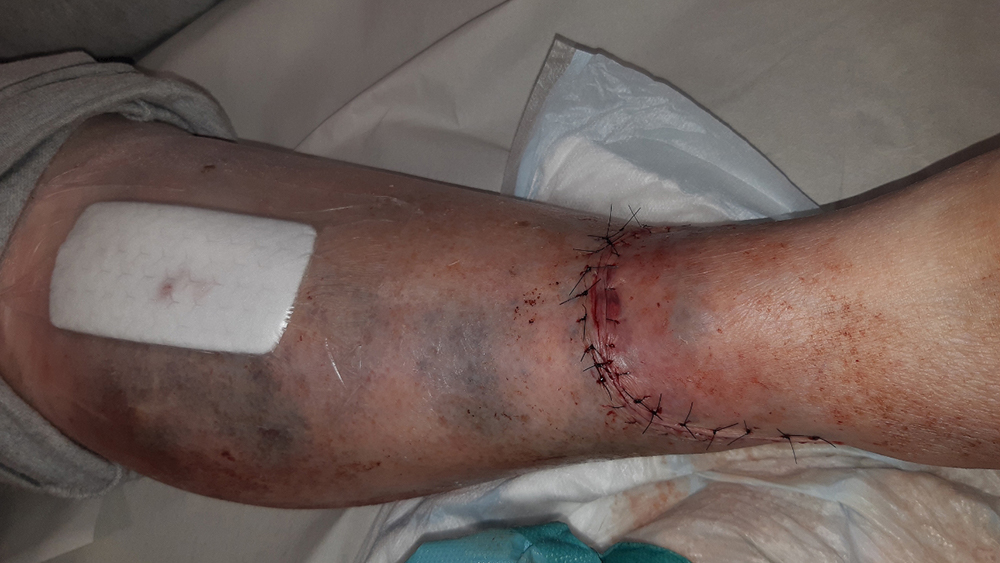
72-year-old female with current treatment for hypertension, peripheral vascular disease and chronic pulmonary disease, presented with a dehisced leg wound.
44 days prior to baseline visit, the patient had fallen, wounding her right ankle, and in addition causing a small contusion in the right leg proximally and anteriorly.
A large hematoma developed in the area of the wound and the skin went into necrosis. The ankle wound was primarily sutured, however, the wound dehisced. Due to the negative progression of the wounds, a revision was performed and a traditional NPWT system was applied. Later, the wounds were found to be still covered with fibrin. There was larval treatment between the wounds care visits, during which time NPWT was stopped.
Treatment regime
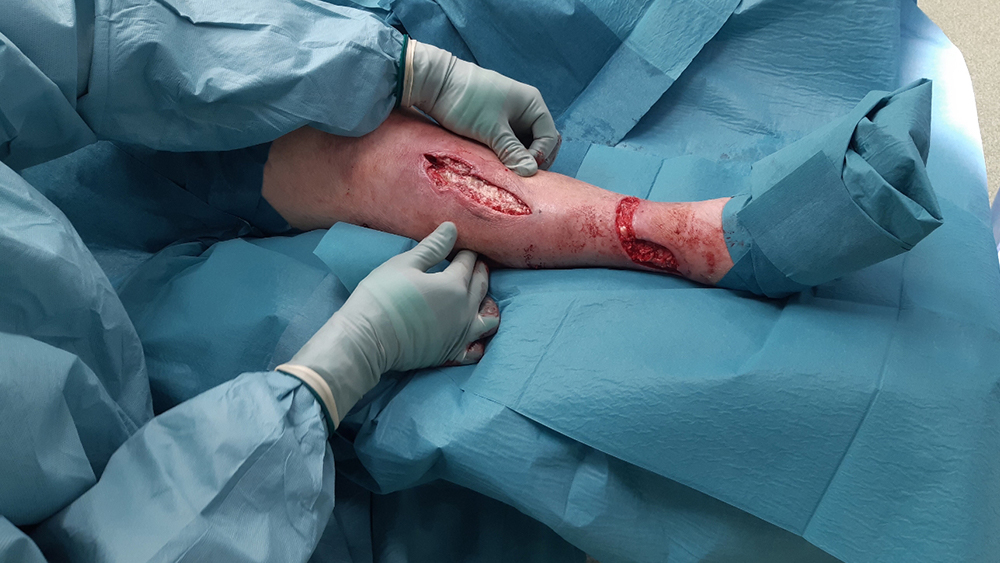
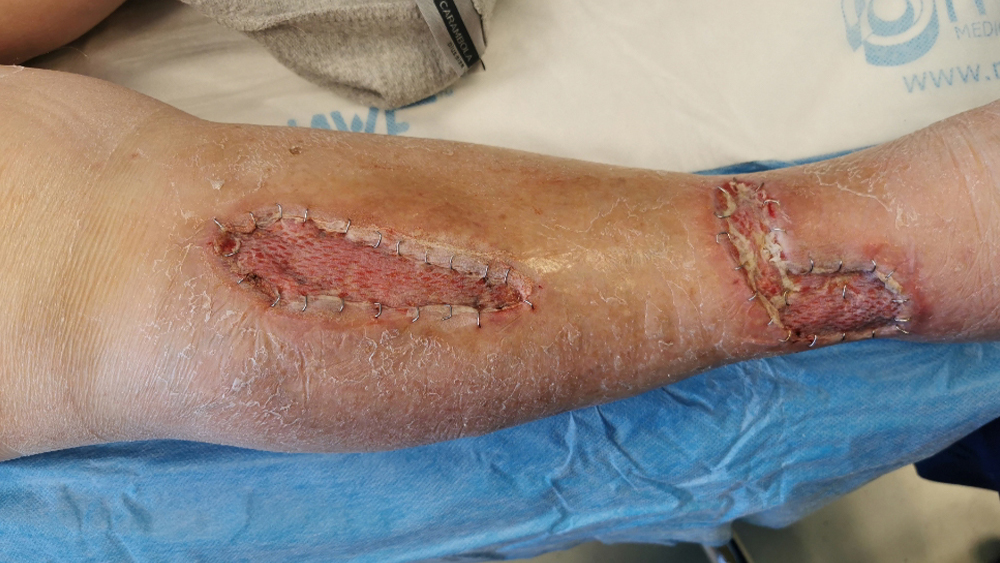
44 days after the trauma an additional wound revision was performed and skin grafts were applied .
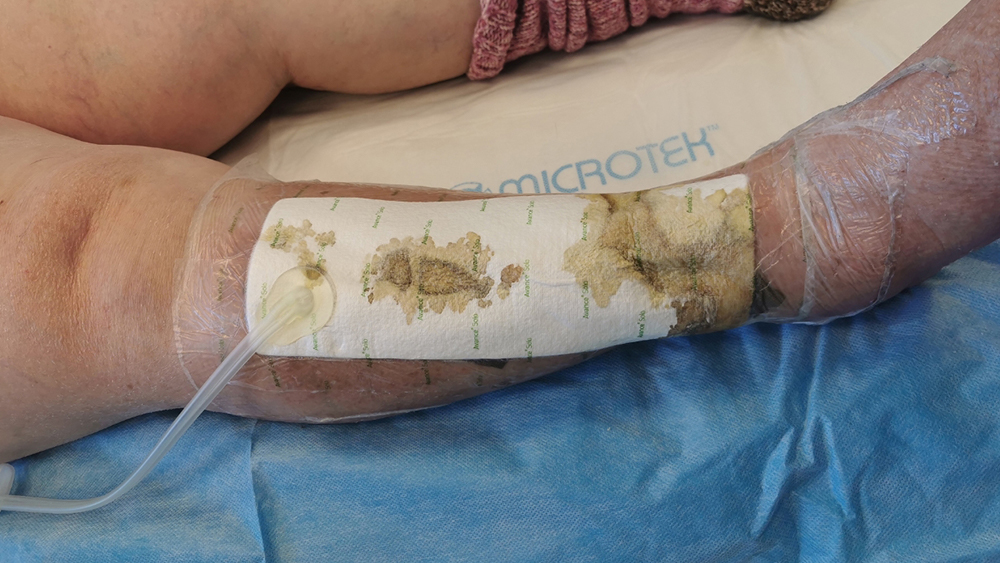
Portable negative pressure wound therapy system was applied successfully under compression bandage and a support stocking. One dressing, size 15 x 30 cm covered both wounds.
Outcome
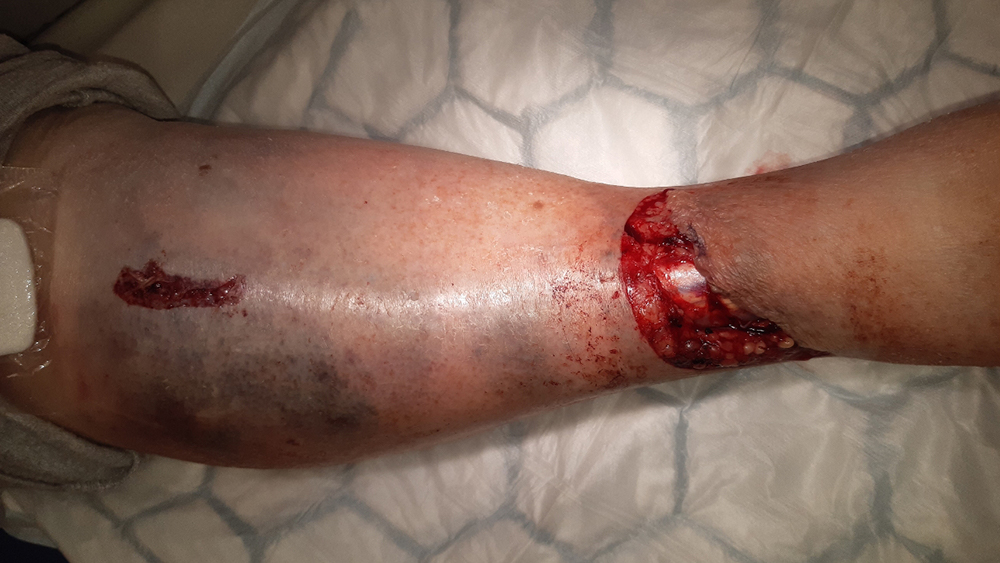
Review at baseline showed two wounds with size 10 cm x 3 cm and 12 cm x 4 cm, respectively both with a depth of 1 cm. The majority of the wound areas were granulated (90%) and there was slough present (10%) but no necrotic tissue. Exudate levels were low and clear/serous in appearance.
12 days after the first visit the wounds had progressed negatively and 80% of the wounds were now epithelialized and 20% still granulated. Levels of exudate in the canister
were low, however the exudate levels around the wounds were medium and brown/bloody as well as viscous in appearance.
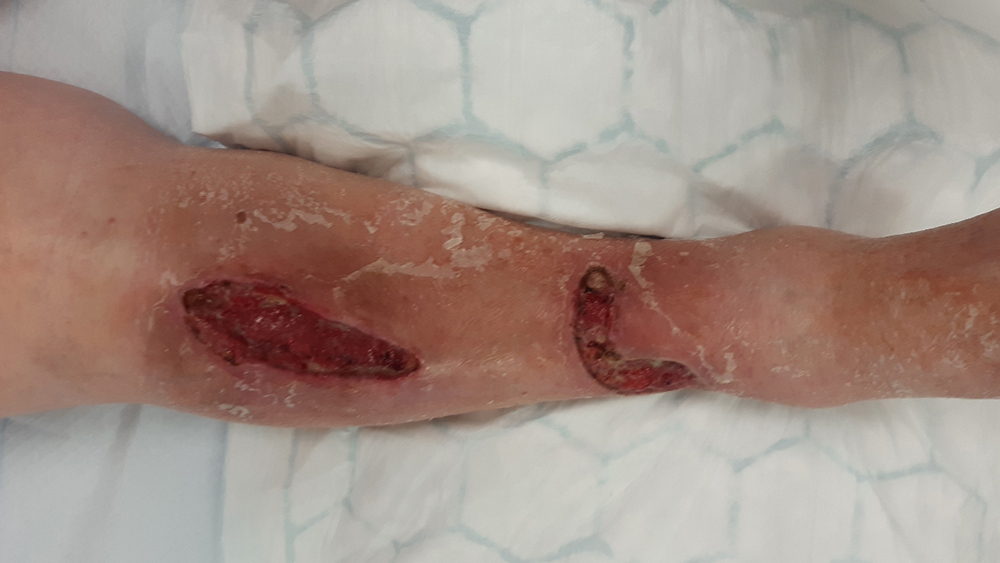
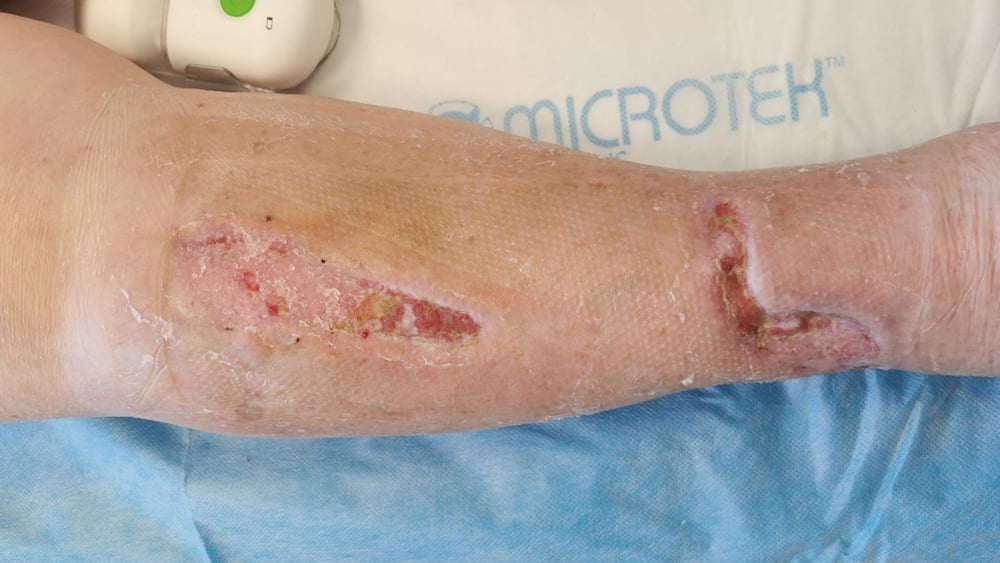
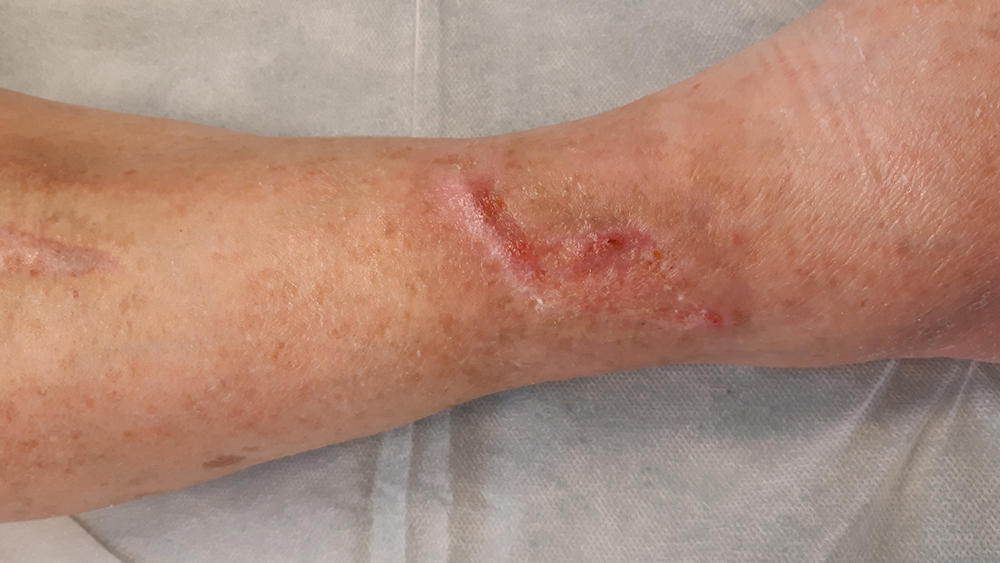
At follow up visit 3 (30 days) the wound on the ankle had decreased to 0,4 cm x 1 cm with a depth of 0.2 cm with 90% of the surface epithelialized and 10% granulated while the second wound was now fully healed.
The exudate was still appearing brown/bloody and viscous and moderate in amount.
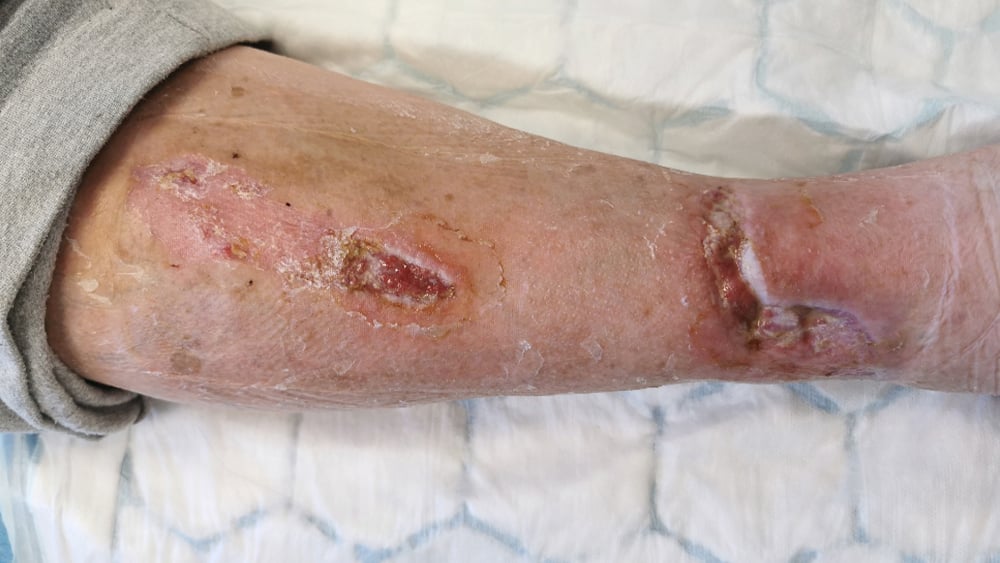
44 days (88 days after the trauma) after first visit both wounds were fully healed.
There was no signs of infection in any of the wounds during the treatment duration and no antibiotics were administered.
The periwound skin appeared continuously healthy the whole period and no wound filler was used.
No additional dressing changes outside of the planned follow-up visits.
Sharp debridement was performed at each follow up visit except visit 4 (44 days after baseline)
The patient reported no pain before or during dressing removal.
Performance
All performance aspects of the device was perceived as ‘Very good’ including suitability of size and shape of the dressing. The dressing size of 15x30 cm formed well on the tibia and covered both skin grafts.
The exudate management ability of the device was good and worked very satisfactory despite one dressing covering a two-wound situation.
The overall impression was very good as the skin graft had improved quickly in the follow-up of a complicated trauma wound followed by revision and skin grafting, and the device had been easy and effortless to use.
Patient experience
The patient was satisfied with the treatment throughout the treatment period, perceiving it as effortless and painless. She was very pleased with the few changes of dressings and easy management of the treatment.